 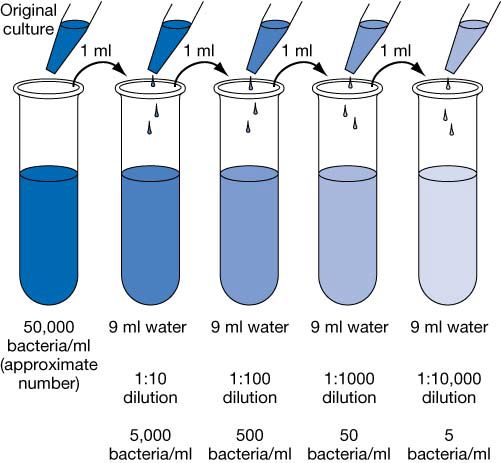
This manual uses a modification of the method of de Man (1983). It is possible to construct many different sets of intervals that satisfy this criterion. The 95 percent confidence intervals in the tables have the following meaning:īefore the tubes are inoculated, the chance is at least 95 percent that the confidence interval associated with the eventual result will enclose the actual concentration. De Man (1983) published a confidence interval method that was modified to make the tables for this appendix. Woodward (1957) recommended that MPN tables should omit those combinations of positive tubes (high for low concentrations and low for high concentrations) that are so improbable that they raise concerns about laboratory error or contamination. Halvorson and Ziegler (1933), Eisenhart and Wilson (1943), and Cochran (1950) published articles on the statistical foundations of the MPN. McCrady (1915) published the first accurate estimation of the number of viable bacteria by the MPN method. In general, this equation can be solved by iteration. T j denotes the number of tubes in the jth dilution. M j denotes the amount of the original sample put in each tube in the jth dilution, G j denotes the number of positive (or growth) tubes in the jth dilution, It is the solution for λ, concentration, in the following equation The MPN is the number which makes the observed outcome most probable. In order to obtain estimates over a broad range of possible concentrations, microbiologists use serial dilutions incubating tubes at several dilutions. The "outcome", i.e., the number of tubes and the number of tubes with growth at each dilution, will imply an estimate of the original, undiluted concentration of bacteria in the sample. The essence of the MPN method is to dilute the sample to such a degree that inocula in the tubes will sometimes but not always contain viable organisms. The individual tubes of the sample are independent. 
Every tube (or plate, etc.) whose inoculum contains even one viable organism will produce detectable growth or change. The bacteria are separate, not clustered together, and they do not repel each other. The bacteria are distributed randomly within the sample. The following assumptions are necessary to support the MPN method. If a confirmation test involves selecting colonies to test, then a statistical adjustment not discussed in this appendix should be used (see Blodgett 2005a.) For simplicity, however, this appendix will speak of these GUs or CFUs as individual bacteria. If, in the microbiologist's experience, the bacteria in the prepared sample in question can be found attached in chains that are not separated by the preparation and dilution, the MPN should be judged as an estimate of growth units (GUs) or colony-forming units (CFUs) instead of individual bacteria. Only viable organisms are enumerated by the MPN determination. McClure in the Bacteriological Analytical Manual (BAM), 7th edition. The following background observations are adapted and extended from the article on MPN by James T. The MPN is particularly useful for low concentrations of organisms (<100/g), especially in milk and water, and for those foods whose particulate matter may interfere with accurate colony counts. Serial dilution tests measure the concentration of a target microbe in a sample with an estimate called the most probable number (MPN). for 10 tubes at 10 ml inocula and link to spreadsheet October 2010: Equation for most probable number (MPN) replaced with graphical version Added expanatory note for the downloadable spreadsheet.April 2015: Contact for this Appendix was updated.Bacteriological Analytical Manual (BAM) Main Pageįor additional Information, contact: Stuart Chirtel or Guodong Zhang 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
